Problem 248. Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrows to indicate the direction of electron flow. Problem 2-49. Calculate Ka values from the following p Ka ‘s: (a) Acetone, p Ka = 19.3. (b) Formic acid, p Ka = 3.75.
Solved 3. Which of the following pairs are not resonance | Chegg.com
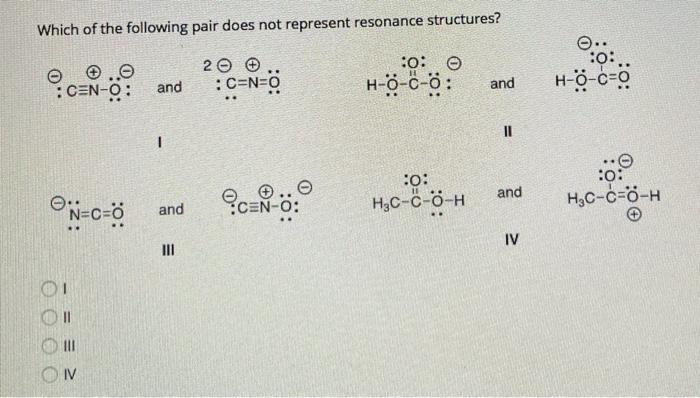
Which of the following pairs of structures does not represent resonating structure :- O clich, and color – OH 0-H 3 ch-C-CH, and X Your Answer @CH=C=0 and EH_C=0.
Source Image: chegg.com
Download Image
8.6: Resonance Structures is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Some molecules have two or more chemically equivalent Lewis electron structures, called resonance structures. Resonance is a mental exercise and method within the Valence Bond Theory of bonding that ….
Source Image: numerade.com
Download Image
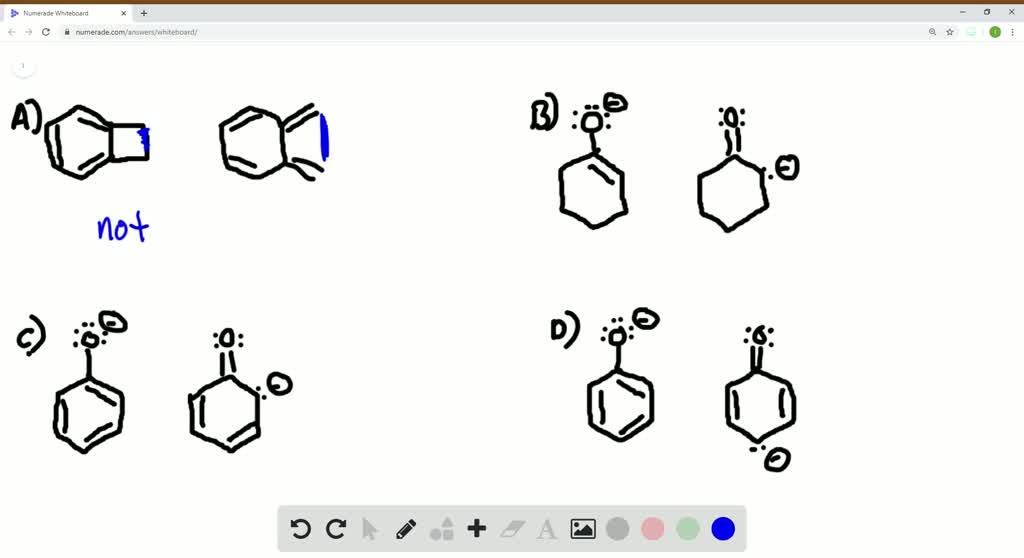
Which of following pairs does not represent resonance sturctures? – YouTube The carbon in contributor C does not have an octet. In general, resonance contributors in which a carbon does not fulfill the octet rule are relatively less important. (rule #1) In structure C, there are only three bonds, compared to four in A and B. In general, a resonance structure with a lower number of total bonds is relatively less important.
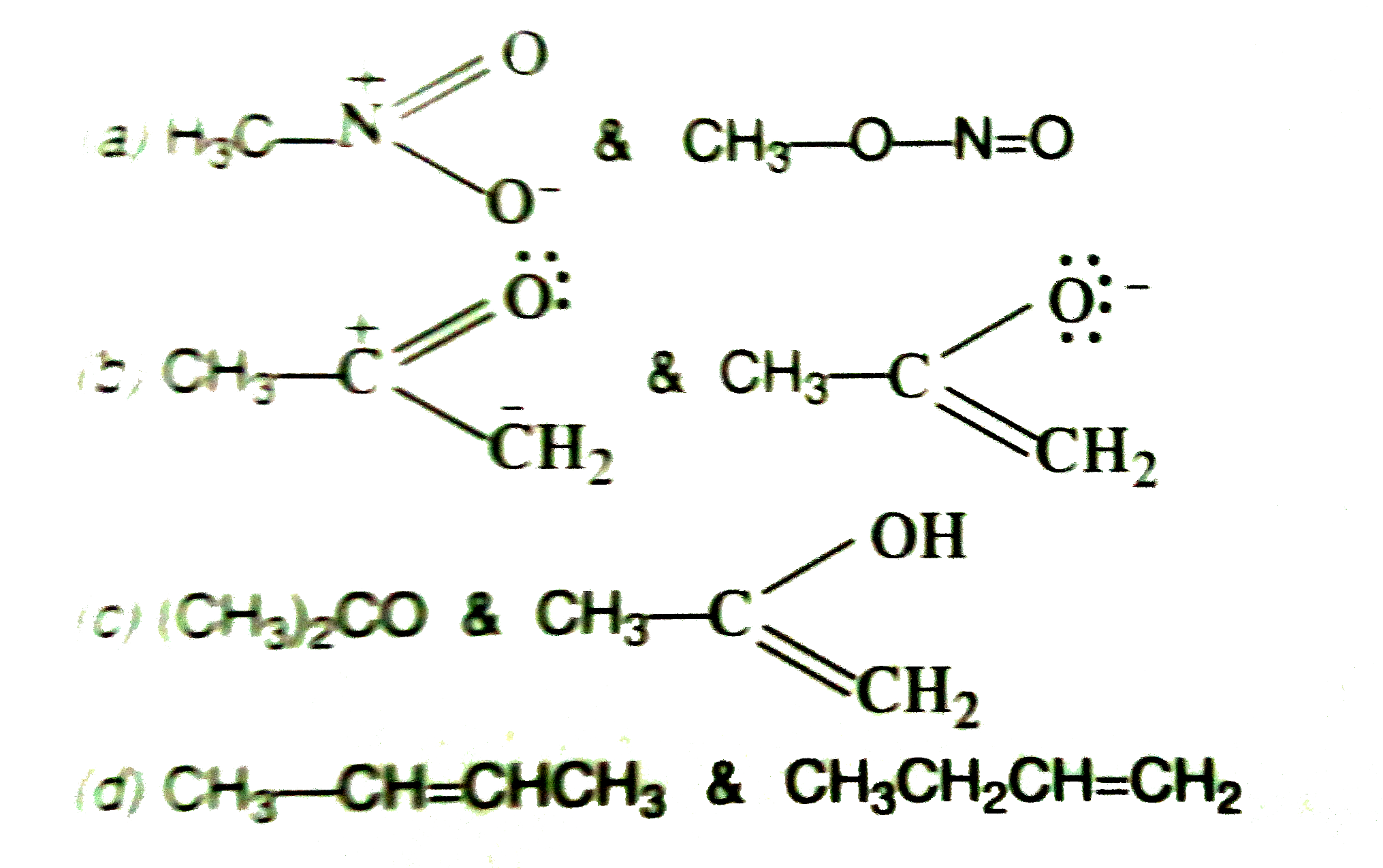
Source Image: doubtnut.com
Download Image
Which Of The Following Pair Does Not Represent Resonance Structures
The carbon in contributor C does not have an octet. In general, resonance contributors in which a carbon does not fulfill the octet rule are relatively less important. (rule #1) In structure C, there are only three bonds, compared to four in A and B. In general, a resonance structure with a lower number of total bonds is relatively less important. Sep 29, 2023The O=C-C: − O=C-C: − grouping is a typical one for which two resonance structures can be drawn. Just as there is a C=O C=O bond to the left of the lone pair, there is a second C=O C=O bond to the right. Thus, we can draw a total of three resonance structures for the 2,4-pentanedione anion.
Which of the following pairs do not constitute resonance structures ?
Question: Which of the following pair does NOT represent resonance structures? 20 :C=N=0 :.:. :CEN-O: and :0 0 H-0-c-: and H-0-c=0 II :: and :: Hoc-C-0-H and Học-cô-H IV 11 III IV Show transcribed image text Here’s the best way to solve it. Expert-verified 100% (16 ratings) In resonating struct … View the full answer Previous question Next question Which of following pairs does not represent resonance sturctures? – YouTube

Source Image: youtube.com
Download Image
Which of following pairs does not represent resonance sturctures? Question: Which of the following pair does NOT represent resonance structures? 20 :C=N=0 :.:. :CEN-O: and :0 0 H-0-c-: and H-0-c=0 II :: and :: Hoc-C-0-H and Học-cô-H IV 11 III IV Show transcribed image text Here’s the best way to solve it. Expert-verified 100% (16 ratings) In resonating struct … View the full answer Previous question Next question
Source Image: doubtnut.com
Download Image
Solved 3. Which of the following pairs are not resonance | Chegg.com Problem 248. Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrows to indicate the direction of electron flow. Problem 2-49. Calculate Ka values from the following p Ka ‘s: (a) Acetone, p Ka = 19.3. (b) Formic acid, p Ka = 3.75.
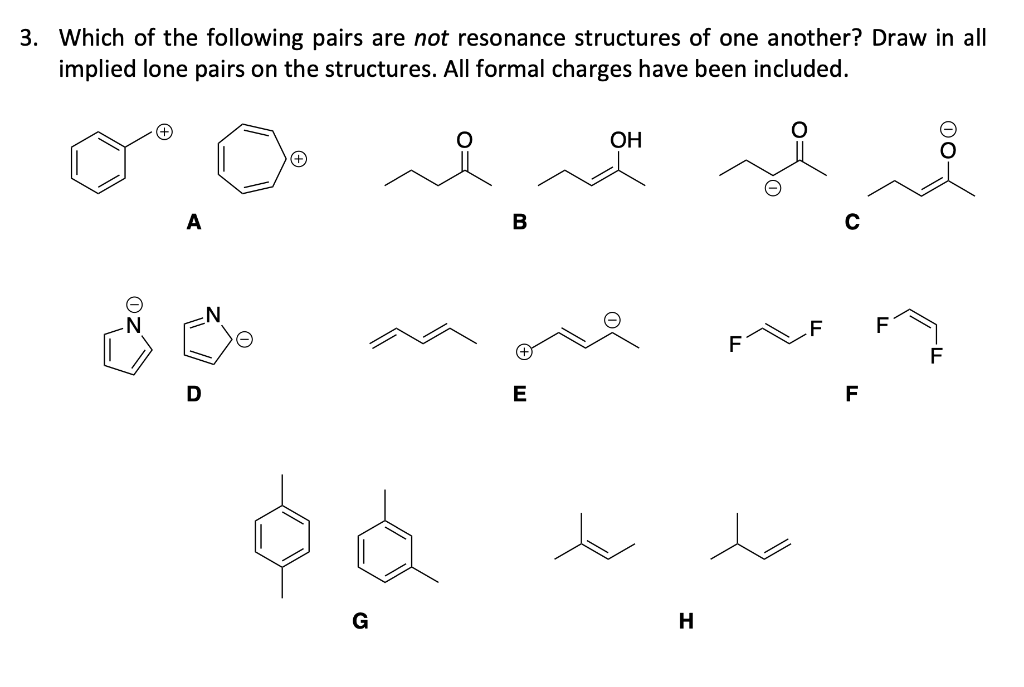
Source Image: chegg.com
Download Image
Which of following pairs does not represent resonance sturctures? – YouTube 8.6: Resonance Structures is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Some molecules have two or more chemically equivalent Lewis electron structures, called resonance structures. Resonance is a mental exercise and method within the Valence Bond Theory of bonding that ….

Source Image: youtube.com
Download Image
Solved Which pair does not represent resonance structures? | Chegg.com Chemistry Chemistry questions and answers Which of the following does not represent a pair of resonance structures? 1 :0: :0: :0: нусн Нусн өсу Нус Сн, НуссH, o 2 О1 This problem has been solved! You’ll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer
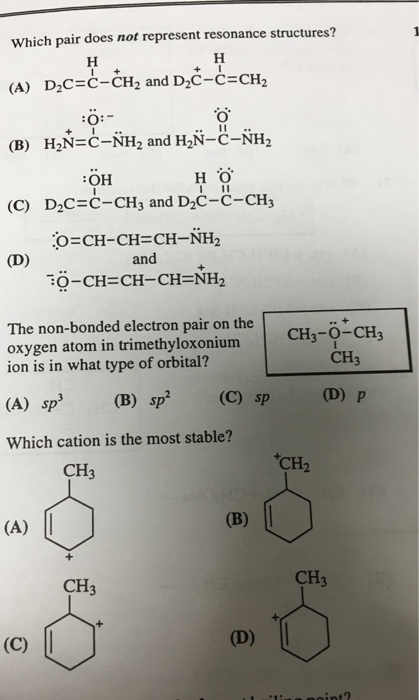
Source Image: chegg.com
Download Image
CH(3)-overset(“^(o+O-H))overset(||)C-CH(3)and CH(3)-underset(o+)overse The carbon in contributor C does not have an octet. In general, resonance contributors in which a carbon does not fulfill the octet rule are relatively less important. (rule #1) In structure C, there are only three bonds, compared to four in A and B. In general, a resonance structure with a lower number of total bonds is relatively less important.
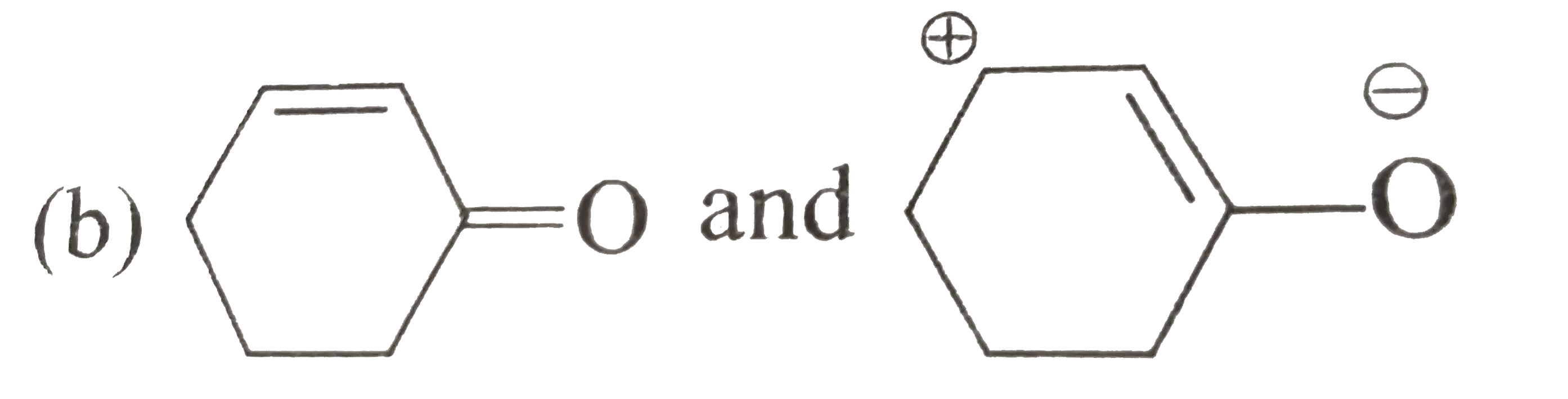
Source Image: doubtnut.com
Download Image
Which of the following pairs do not constitute resonance structures ? Sep 29, 2023The O=C-C: − O=C-C: − grouping is a typical one for which two resonance structures can be drawn. Just as there is a C=O C=O bond to the left of the lone pair, there is a second C=O C=O bond to the right. Thus, we can draw a total of three resonance structures for the 2,4-pentanedione anion.

Source Image: doubtnut.com
Download Image
Which of following pairs does not represent resonance sturctures?
Which of the following pairs do not constitute resonance structures ? Which of the following pairs of structures does not represent resonating structure :- O clich, and color – OH 0-H 3 ch-C-CH, and X Your Answer @CH=C=0 and EH_C=0.
Which of following pairs does not represent resonance sturctures? – YouTube CH(3)-overset(“^(o+O-H))overset(||)C-CH(3)and CH(3)-underset(o+)overse Chemistry Chemistry questions and answers Which of the following does not represent a pair of resonance structures? 1 :0: :0: :0: нусн Нусн өсу Нус Сн, НуссH, o 2 О1 This problem has been solved! You’ll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer